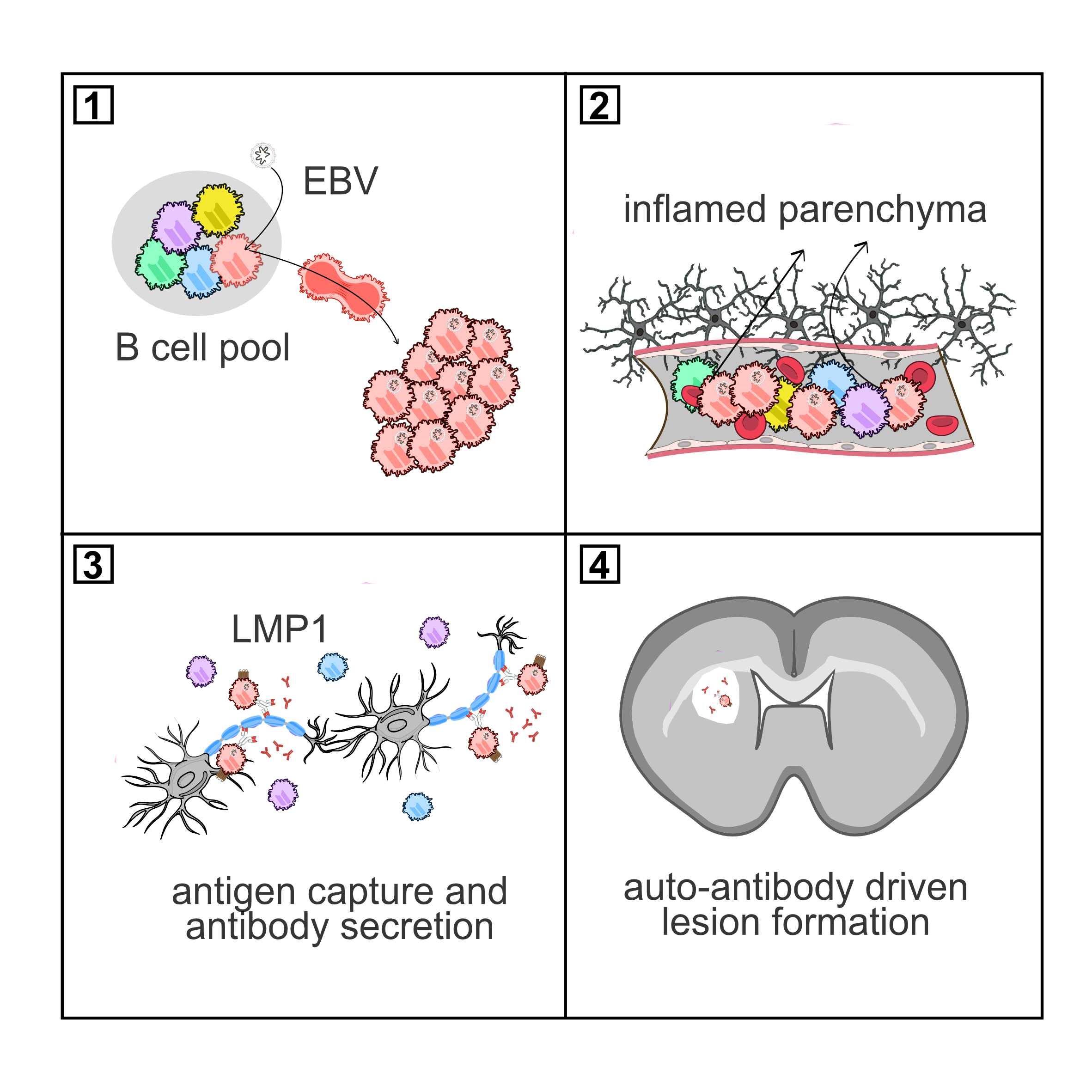
A new publication from Research Center for Clinical Neuroimmunology and Neuroscience Basel (RC2NB), led by Tobias Derfuss team, proposes a clear mechanistic explanation for the long-observed link between EBV and MS:
the connection between Epstein-Barr virus (EBV) and multiple sclerosis. This connection is well established epidemiologically and has become a focus of MS research internationally, but no clear model has emerged that can explain mechanistically why only EBV, and not some other virus is a necessary prerequisite for the disease. Researchers associated with the RC2NB led by Professor Tobias Derfuss have focused on the ability of EBV to protect B cells that are activated by self antigens. In a physiological immune response, such as against a virus, a B cell is activated by encounter with antigen, and once activated can only survive for a few hours, unless it receives survival signals from T cells that recognize the same antigen. The paper shows how myelin-reactive B cells that enter the central nervous system normally die because of the absence of this T-cell-derived survival signal. However, if such CNS-infiltrating, myelin-reactive B cells express the EBV-encoded protein LMP1, this provides a substitute signal, and enables the B cells to survive. The surviving B cells can produce antibody, and in this leads to localized demyelination reminiscent of the lesions seen in MS.
These basic immunological mechanisms were demonstrated in animal models, and to assess the plausibility of this model in humans, the team confirmed that myelin-reactive B cells are present in the naive repertoire of healthy people, and also that LMP1-expressing B cells are detectable in lesions in the brains of patients with MS.
This new model has important implications for the prevention and treatment of the disease. Because CNS infiltration by EBV-infected, myelin-reactive B cells is only statistically likely in the presence of a large expanded clone, the model is consistent with the association of severe EBV infections in adolescence with later MS incidence, and predicts that a partially effective vaccine, that failed to prevent infection, but limited the extent of viral replication would still be effective in preventing MS. Regarding treatment, the same requirement for a large population of circulating infected B cells of a particular specificity predicts that complete eradication of peripheral mature B cells with CD20-depleting antibodies ought to be curative, rather than merely suppressing disease for the duration of depletion.
Full article here: https://www.sciencedirect.com/science/article/pii/S0092867425014801

Figure legend: Graphical summary of the conclusions of the study. In this model, early EBV infection leads to the random expansion of a clone of myelin-reactive, latently infected B cells (1). Later in life (2) an insult such as a brain infection causes immune cell influx (3), including B cells from the myelin-reactive clone. Expression of the viral protein LMP1 enables these B cells to escape tolerance, secrete antibody, and cause localized demyelination.